- No products in the cart.
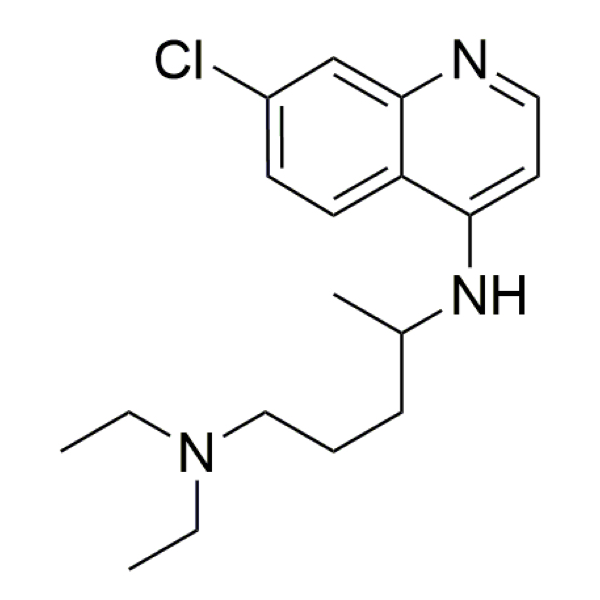
Chloroquine – CAS 54-05-7
€100 – €150
Chloroquine is provided by Santiago Lab. It is one of the 5 compounds recommended by WHO for the investigation of the treatment of COVID-19. You can order chloroquine individually or as a set of 5 antivirotics recommended by WHO.
Purity (LC-MS)
99 % | Certificate of Analysis
Package contents
Chloroquine sulfate
This compound is for research use only. We do not sell to patients.
| Size | Availability | Price | Quantity |
|---|---|---|---|
| 100 mg | In stock |
€100 | |
| 1 g | In stock |
€150 |
Characterisation
CAS: 54-05-7
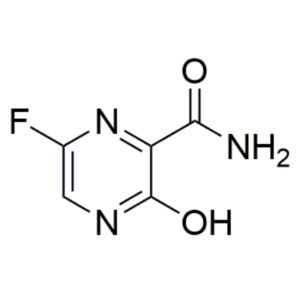
IUPAC Name: 4-N-(7-chloroquinolin-4-yl)-1-N,1-N-diethylpentane-1,4-diamine
Other names: Aralen; Chingamin; Nivaquine; Chlorochin; Chloroquine
Molecular weight: 319.88 g/mol
Molecular formula: C18H26ClN3
Description
Chloroquine is a medication which is used to prevent and treat malaria in areas where malaria remains sensitive to its effects. Certain types of malaria, resistant strains, and complicated cases typically require different or additional medication. It is also occasionally used for amebiasis that is occurring outside the intestines, rheumatoid arthritis, and lupus erythematosus. Chloroquine is also being studied for the treatment of COVID-19.
Chloroquine is a member of the drug class 4-aminoquinoline and it was discovered in 1934, by chemist Hans Andersag and coworkers at the Bayer laboratories, who named it Resochin. Chloroquine has also significant antiviral effects. It increases late endosomal and lysosomal pH, resulting in the impaired release of the virus from the endosome or lysosome – the release of the virus requires a low pH. The virus is therefore unable to replicate (releasing its genetic material into the cell).
Chemicals are distributed worldwide
Buy Chloroquine now, get your order in 48 hours
- Shipping through DHL in 48 hours
- All compounds are safely and rigorously packed
Payment
- We are sending the invoice the same day as the shipment
- We are able to modify the invoice for the academic institution, so the order can be paid from grants
| References | 1. Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Research 2020, 30 (3), 269-271. |
|---|
Related products:
-
Remdesivir – CAS 1809249-37-3
€200 – €5900 -
Hydroxychloroquine – CAS 118-42-3
€250 – €400 -
Azithromycin – CAS 83905-01-5
€100 – €200 -
Favipiravir – CAS 259793-96-9
€200 – €350 -
Aviromix
€700