- No products in the cart.
Reagent of the month – CBS oxazaborolidine
The Corey-Bakshi-Shibata (CBS) catalyst is an organocatalyst that is used in asymmetric ketone reductions (Corey-Itsuno reduction). The structure of the CBS oxazaborolidine catalyst is shown in Figure 1.
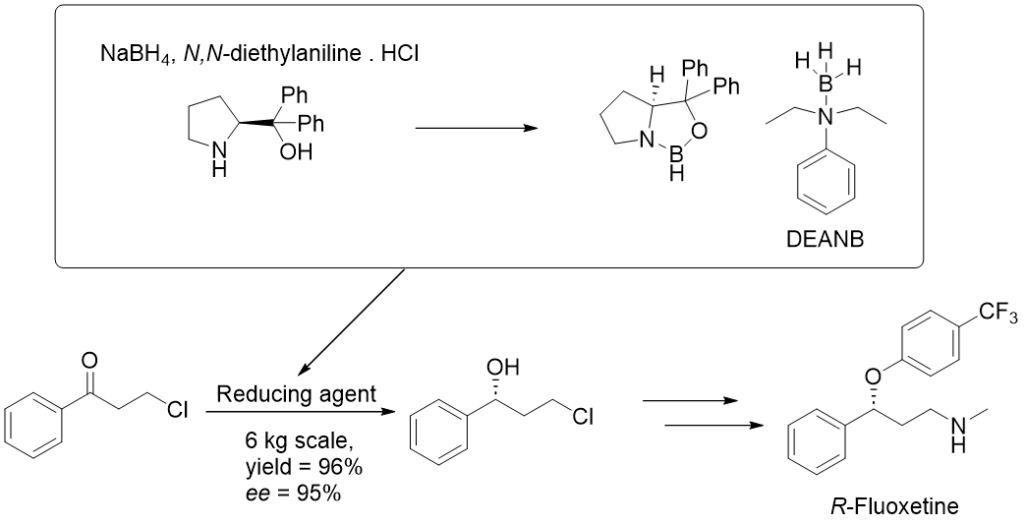
Preparation of the CBS catalyst
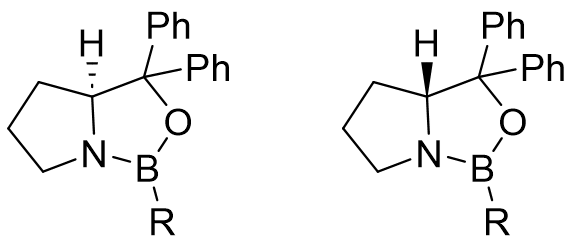
CBS oxazaborolidine can be synthesized by reacting the corresponding enantiomer of diphenylprolinol with borane and then alkylating it, or by reacting it with boroic anhydrides. Diphenyl prolinol can be synthesized from the corresponding proline enantiomer. The catalyst’s active form is then formed in-situ by the coordination of borane with the lone electron pair of nitrogen (Figure 2.). The quantitative reducing agent is borane complexes with THF, dimethyl sulfide, or various amines.
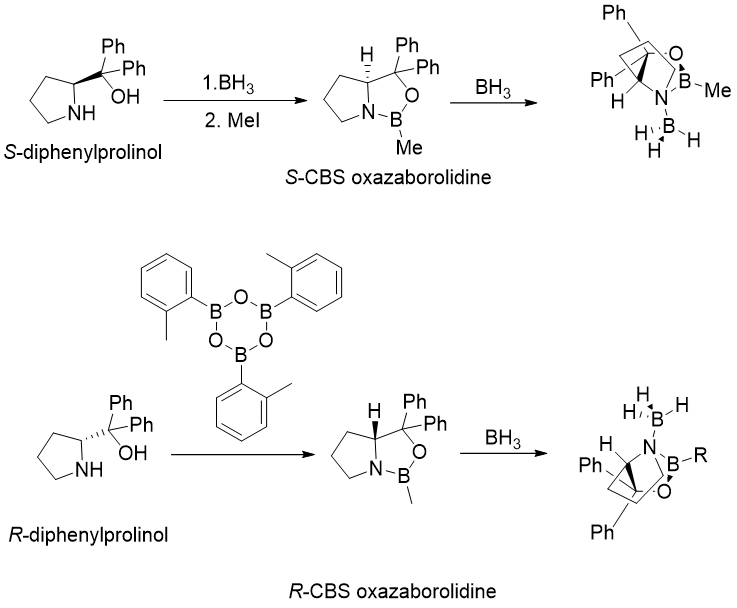
Enantioselectivity of the Corey–Itsuno reduction
The Corey-Itsuno reduction is most useful for ketones with two substituents that differ significantly in size. Because of the large difference in the steric bulk of the methyl and phenyl substituents, acetophenone is reduced with very high ee. The larger ketone substituent (RL) in the transition state is oriented away from the R substituent on boron in the CBS catalyst, while the smaller substituent (RS) is closer to the R substituent (Figure 3.). The R substituent can be simply hydrogen, methyl, butyl, phenyl, and so on, and is chosen for each substrate to achieve the best enantioselectivity.
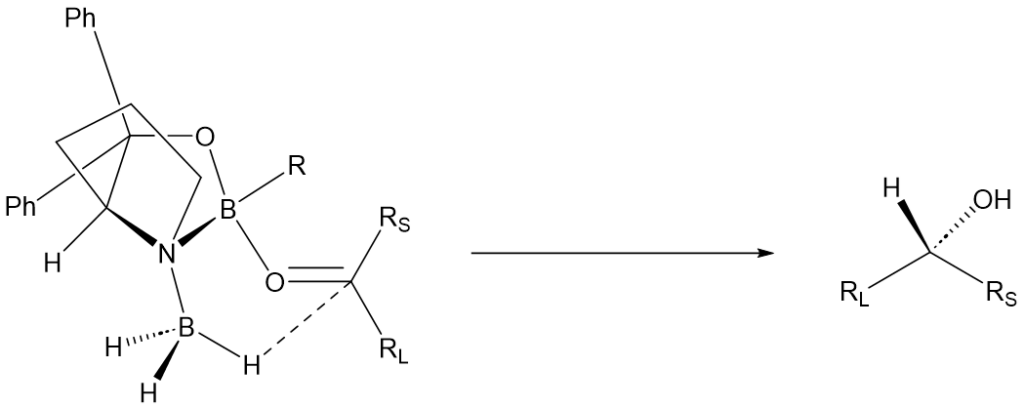
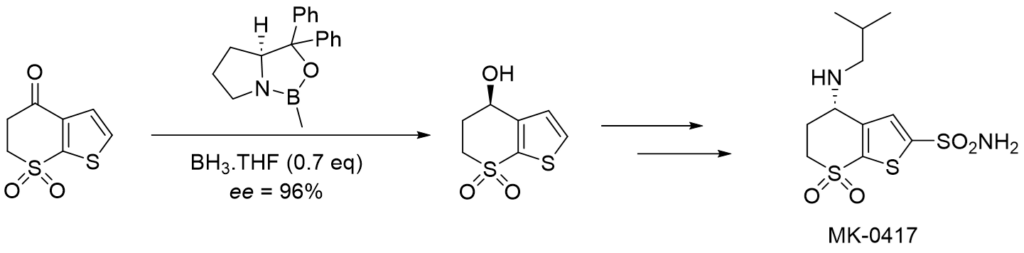
Application of the CBS catalyst in synthesis
Corey–Itsuno reduction was first discovered by the Itsuno group in the 1981 (doi:10.1039/C39810000315) and further developed by Corey, Bakshi and Shibata in 1987 (doi:10.1021/ja00252a056). Since then, it has been used in numerous total syntheses of natural products as well as large-scale drug syntheses. It can be used for example, in one step of the synthesis of the carbonic anhydrase inhibitor MK‑0417 developed by Merck and later marketed as Dorzolamide under a brand name Trusopt as an eye drop medication for the treatment of ocular hypertension (Figure 4.). The reaction is sensitive to water which negatively impacts the enantioselectivity, so it has to be performed in dry solvents (doi:10.1021/jo00002a050).
In 2012, an easy and scalable one-pot procedure for producing the asymmetric reducing agent was developed by combining NaBH4 with diethylaniline hydrochloride and diphenylprolinol. This is an improvement over sensitive and hazardous borane complexes. It was demonstrated in kilogram scale for the synthesis of numerous drug precursors for commercially available drugs such as fluoxetine, dapoxetine, rivastigmine, and others.(doi.org/10.1021/op300034u) (Figure 5).