- No products in the cart.
Reagent of the month – DDQ and quinone-derived oxidants
Reagents derived from p-quinone such as 2,3-dichloro-5,6-dicyanobenzoquinone (DDQ) or p-chloranil are used in a wide range of applications in organic synthesis as quantitative oxidants, however, numerous quinone catalysed transformations using other chemicals or electrodes as quantitative oxidants have recently been developed (https://doi.org/10.1002/anie.201505017)
Synthesis
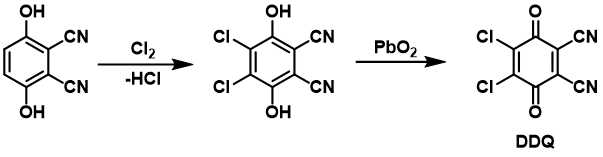
DDQ can be made by chlorinating 2,3-dicyanohydroquinone and then oxidising it with lead (IV) oxide.
Applications of DDQ in the synthesis
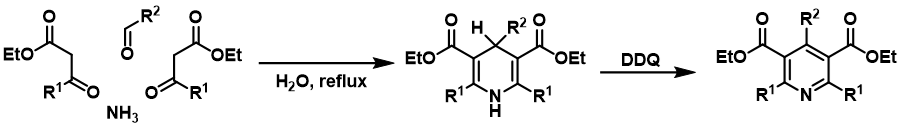
DDQ is probably best known for its ability to dehydrogenate hydroaromatic compounds to their aromatic analogues, which is used as the final step in many heterocyclic aromatic compound syntheses (e. g. Hantzsch synthesis of pyridine derivatives), but it can also be used for selective oxidation of alcohols and ethers in benzylic or allylic positions to corresponding aldehydes or ketones (https://doi.org/10.1002/).
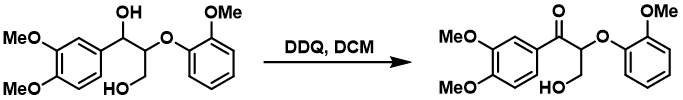
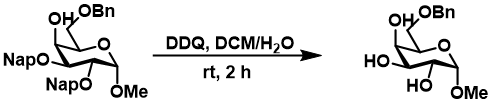
This oxidation is destructive in the case of ethers and works better on more activated benzyl derivatives. In wet DCM can thus be used to selectively deprotect Nap (2-naphytlymethyl) or PMB (p-methoxybenzyl) groups while leaving benzyl groups untouched, which is commonly used in carbohydrate chemistry (https://doi.org/10.1016/S0040-4020(01)90593-9; https://doi.org/10.1016/S0040-4039(01)00563-9).
DDQ is used in a variety of oxidative C-C Scholl type coupling reactions on both aromatic and aliphatic substrates. Cyclotrimerization of substituted benzenes (https://doi.org/10.1016/j.tetlet.2014.12.141) and many intramolecular cyclizations (https://doi.org/10.1021/jo401807x) are examples of such coupling.
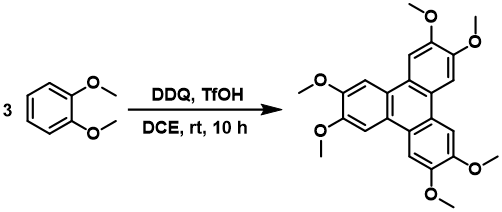
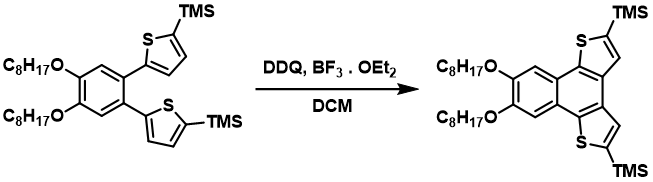
Industrial applications of DDQ
DDQ is used in numerous syntheses of pharmaceuticals for dehydrogenation of hydroaromatic intermediates. The dehydrogenation step in the synthesis of the anti-obesity and cardiovascular disease prevention drug Rosuvastatin is one example of such use. S-methylisothiourea is first condensed with ethyl 2-(4-fluorobenzylidene)-4-methyl-3-oxopentanoate, and the resulting 1,2-dihydropyrimidine derivative is aromatized by dehydrogenation with DDQ. (https://doi.org/10.1016/S0968-0896(96)00248-9)
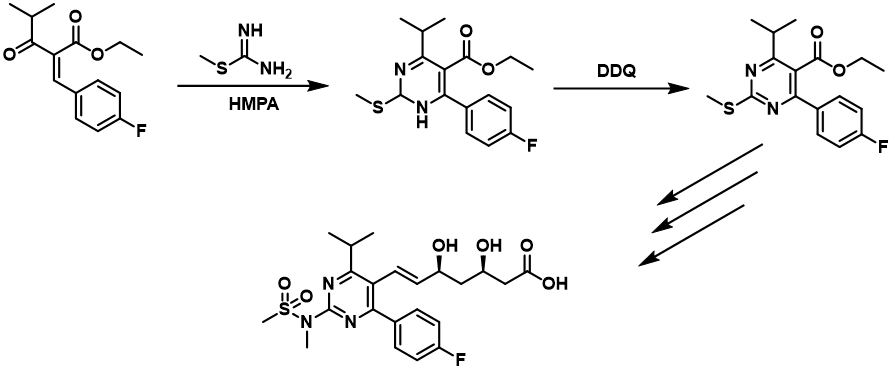
We have a lot of experience at Santiago lab with various oxidation reagents (such as DDQ) for performing difficult syntheses. If you want to learn more about our services, discuss specific research or custom synthesis project, or receive a proposal, please contact Krystof Sigut at krystof.sigut@santiago-lab.com
or reach him on the phone +420 776 750 591.


