- No products in the cart.
Introduction to antibody-drug conjugates (ADCs)
The selectivity of therapeutic drugs to cancer cells over healthy tissue is one of the most challenging difficulties in cancer treatment. The employment of sophisticated drug delivery systems is a modern answer to this challenge. This strategy has been used in the production of antibody-drug conjugates (ADCs), which are therapeutic medicines that combine monoclonal antibody targeting with highly potent cytotoxic chemicals. This combination results in current chemotherapeutics with high selectivity over normal cells and low systemic toxicity.
What are they made of?
In general, antibody-drug conjugates (ADC) is made up of three major components (Fig. 1). The first is a monoclonal antibody, which, as previously said, is responsible for selective targeting. These antibodies are designed to attach to the surface receptors on cancer cells and are tailored to certain types of cancer cells in various tumours. Additionally, antibodies must be non-immunogenic and capable of internalization into cancer cells after attaching to the antigen.
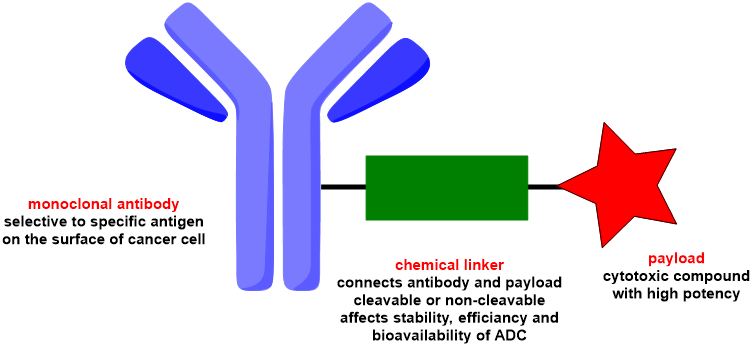
Fig. 1: Schematic representation of antibody-drug conjugate (ADC).
The cytotoxic agent, often known as the payload, is another component of antibody-drug conjugate (ADC). The payload must meet several criteria, including high cytotoxic potency (ideally sub-nanomolar), chemical stability before reaching the target, the presence of a suitable functional group in its structure that allows derivatization, and, most importantly, its potency must be retained after cleavage inside the target cell.
Most payloads are derived from natural substances and are either microtubule disruptors (e.g., auristatins, maytansinoids) or DNA damaging agents (e.g., calicheamicins, duocarmycins, camptothecins, or doxorubicin) (Fig. 2).
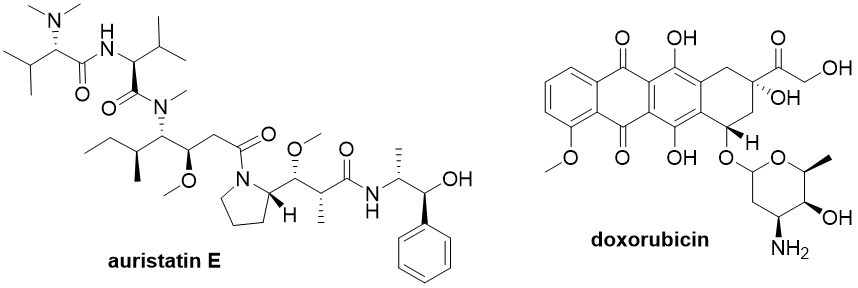
Fig. 2: Structures of microtubule disruptor auristatin E and DNA damaging agent doxorubicin.
The third component of is the linker, which connects the antibody and the payload. The linker chosen is critical since it influences the performance and attributes of the finished ADC. It must be systemically stable so that the antibody-drug conjugate (ADC) is not cleaved before reaching its destination.
Furthermore, the linker must not interfere with antigen binding and must release the active payload when required. Both non-cleavable and cleavable linkers can be employed, but the latter is preferred. Cleavable linkers can be pH sensitive or enzymatically labile, adding another level of selectivity and control over payload release.
Mechanism of action of antibody-drug conjugates (ADCs)
All of the above-mentioned qualities are critical for the desired effect of antibody-drug conjugates (ADCs). Its method of action begins with the binding of antibody-drug conjugate (ADC) via the antibody component to a specific antigen on the surface of a cancer cell (Fig. 3). Following internalization, the whole complex is broken and destroyed in the lysosome, followed by the release of active cytotoxic payload inside the cell. Cytotoxic damage results in apoptosis and the death of cancer cells.
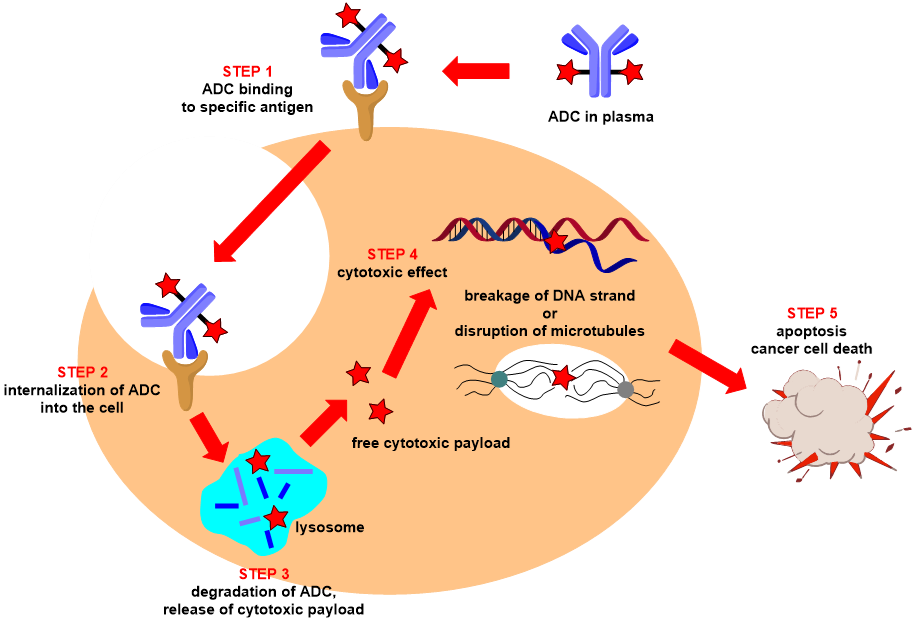
Fig. 3: Schematic depiction of the mechanism of action.
Conclusion
Antibody-drug conjugates (ADCs) have emerged as a potent class of oncology therapeutic agents, with over ten FDA-approved medications (12 ADCs available through 2022) on the market. Even though the concept of ADCs is very simple and understandable, the successful design and synthesis of effective and properly functioning antibody-drug conjugates (ADCs) are pretty tricky. We will return to this subject, as this is not our final essay on antibody-drug conjugates (ADCs)!
Santiago lab offers various services for your research. If you are interested, you can read more about our custom synthesis and contract research.
References
Khongorzul, P.; Ling, C. J.; Khan, F. U.; Ihsan, A. U.; Zhang, J. Antibody–Drug Conjugates: A Comprehensive Review. Mol Cancer Res 2020, 18 (1), 3–19. https://doi.org/10.1158/1541-7786.MCR-19-0582
www.adcreview.com/the-review/antibody-drug-conjugates/what-are-antibody-drug-conjugates/
Jain, N.; Smith, S. W.; Ghone, S.; Tomczuk, B. Current ADC Linker Chemistry. Pharmaceutical Research 2015, 32, 3526–3540. https://doi.org/10.1007/s11095-015-1657-7
Tsuchikama, K.; An, Z. Antibody-drug conjugates: recent advances in conjugation and linker chemistries. Protein & Cell2018, 9, 33–46. https://doi.org/10.1007/s13238-016-0323-0


