- No products in the cart.
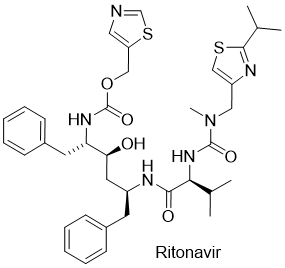
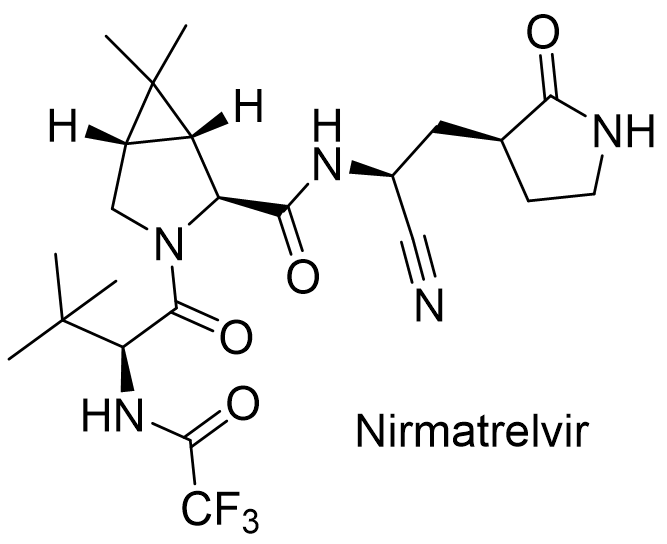
Paxlovid (Nirmatrelvir/Ritonavir)
Paxlovid is a new potential drug for COVID-19 invented by Pfizer. Paxlovid is composed of two active ingredients, Nirmatrelvir and Ritonavir. Similarly, a mixture of Lopinavir and Ritonavir drugs is already used against HIV. Currently, there are big expectations that Paxlovid can replace widely used Remdesivir (developed by Gilead Sciences) and his biologically active Remdesivir triphosphate in the treatment of COVID-19.
Mechanism of action
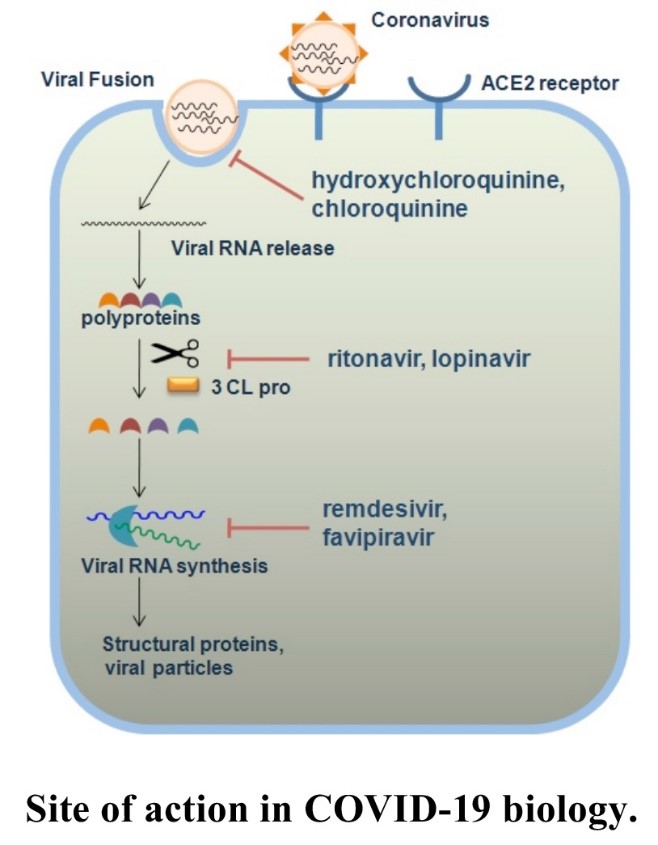
During viral replication, long polypeptides are synthesised, which must then be fragmented into essential viral proteins. Particular proteases (Mpro) are involved in this process, and a possible antiviral effect is actually the inhibition of these proteases. However, protease inhibitors are often metabolised very rapidly in the human body, and therefore, they have to be used in high doses. This results in a higher frequency of side effects. One possible way protease inhibitors are metabolised is through the cytochrome P450 3A4-CYP3A4.
History and current state of Paxlovid
Ritonavir is a drug approved in 1996 which is widely used, not exclusively, to treat HIV/AIDS. Ritonavir selectively binds to and inhibits cytochrome P450 3A4, thereby prolonging the effect of protease inhibitors. Thus, Ritonavir alone does not have an antiviral effect but is used to extend the impact of protease inhibitors.
Another component of Paxlovid, Nirmatrelvir (PF-07321332) is a new potential drug developed by Pfizer. It inhibits the SARS-CoV-1 virus protease (Mpro), which fragments long protein chains into the parts the virus needs to reproduce. Nirmatrelvir acts as a substrate for the virus protease in the cell, thereby deceiving the virus and blocking its replication.
According to study results, Paxlovid (Nirmatrelvir/Ritonavir) reduced hospitalisation or death by 89% for 1,219 patients when given within three days of symptom onset. Pfizer is currently awaiting approval from European and US drug agencies.
Santiago Lab and Antiviral research
At Santiago Lab we are constantly working on antiviral drug research and you can read about it in our articles. Many of our prepared antiviral compounds are commercially available through our e-shop. All our products are lyophilized to ensure long shelf life and the best stability during shipment (usually on dry ice to most countries worldwide).
If you would like to know more, or we can help you with your research or synthesis, write an email to Krystof Sigut on krystof.sigut@santiago-lab.com or reach him on the phone +420 776 750 591.
Drop us a line
We will respond as soon as possible, typically within 48 hours.
Paxlovid references
https://www.nature.com/articles/d41573-021-00202-8
https://academic.oup.com/jac/article/53/1/4/681028?login=true
https://cen.acs.org/acs-news/acs-meeting-news/Pfizer-unveils-oral-SARS-CoV/99/i13
https://www.frontiersin.org/articles/10.3389/fpubh.2020.00365/full


