- No products in the cart.
Reagent of the month – November – Langlois reagent
Trifluormehylation is an effective technique to increase drug candidate solubility and lipophilicity, and so strategies for adding the CF3 group are being investigated. The Langlois reagent (sodium trifluormethylsulfinate) is a valuable reagent for radical trifluormethylation of a wide range of substrates. Bernard R. Langlois’ group first developed methodology for radical trifluormethylation of arenes with optional Cu2+ catalysis in 1991 (https://doi.org/10.1016/0040-4039(91)80524-A), but since then, numerous trifluormethylation methods of other substrates have been developed (https://doi.org/10.1002/adsc.201400370).
Preparation of Langlois reagent
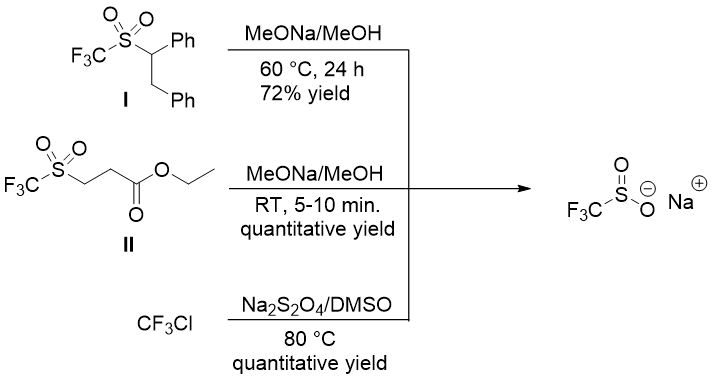
Langlois reagent can be prepared by elimination of trifluormethylsulfones such as I or II by sodium methoxide, or by reacting CF3Cl with sodium dithionate (Na2S2O4) (Figure 1).
Similarly, alternative perfluoralkylation agents can be synthesized utilizing other perfluoralkylsulfones. Perfluoralkylation processes of heteroarenes were successfully carried out using reagents such as C2F5SO2Na, C3F7SO2Na, C6F13SO2Na or C8F17SO2Na (https://doi.org/10.1002/adsc.201300199).
Applications in synthesis
When compared to other trifluormethylation agents such as electrophilic (CF3+ producing) Togni’s or Umemoto’s reagents or nucleophilic reagents such as Ruppert’s reagent (CF3SiMe3), Langlois reagent has a wider substrate scope due to its nucleophilic or electrophilic character.
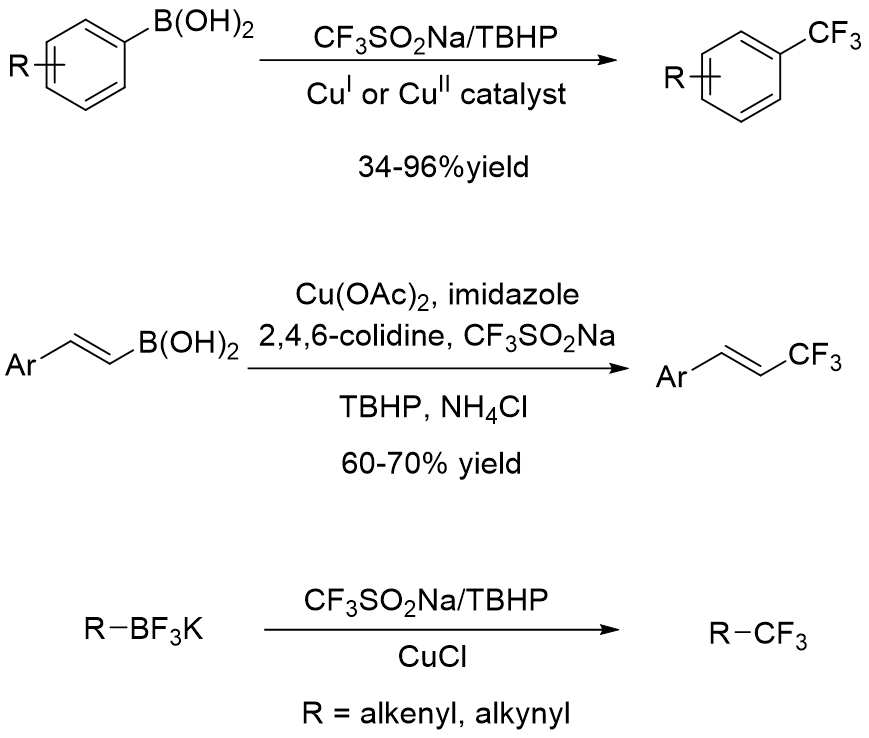
In the presence of an oxidant such as AgOTf, tertbutylhydroperoxide (TBHP), or Mn(OAc)3, trifluormethanesulfinate produces a CF3 radical, which can be used for addition or substitution reactions on aryl or vinylboronic acids and potassium organotrifluorborates (Figure 2) (https://doi.org/10.1021/ol3022726, https://doi.org/10.1039/C2CC36554E, https://doi.org/10.1021/jo4023233), arenes and alkenes (Figure 3) (https://doi.org/10.1039/C3CC41667D, https://doi.org/10.1002/anie.201303576).
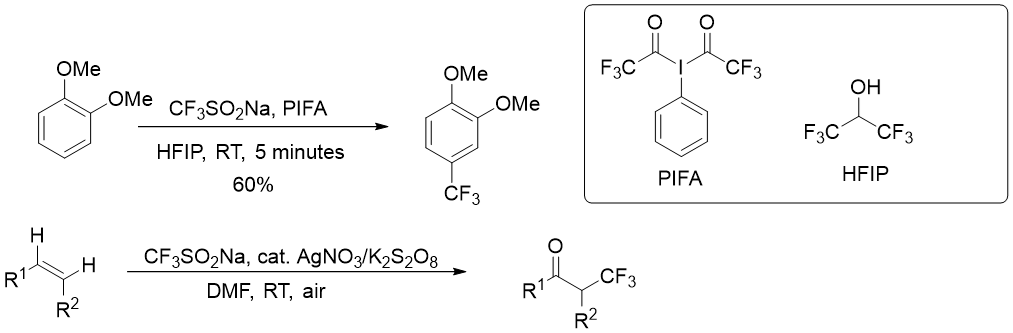
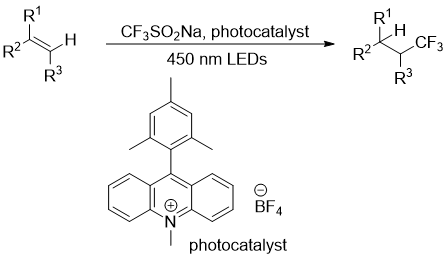
Oxotrifluormethylation of alkenes can be done under oxidative circumstances, but a superior photochemical technique for hydrotrifluormethylation was devised in 2013 (Figure 4) (https://doi.org/10.1039/C3SC51209F).
Langlois reagent in drug synthesis
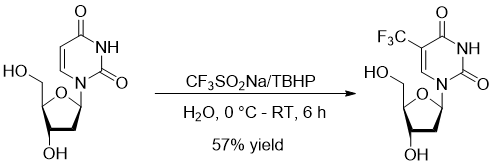
Phil S. Baran’s group described direct C-H trifluormethylation of deoxyuridine with Langlois reagent in 2011 (10.1073/pnas.1109059108), providing Trifluridine – an antiviral drug against herpes virus in a moderate yield in aqueous solution, making it possible to produce this drug using greener chemistry (Figure 5).