- No products in the cart.
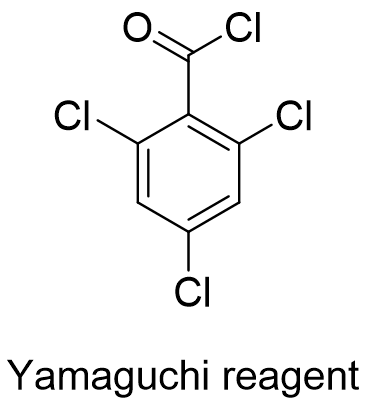
Yamaguchi reagent – Reagent of the month June
2,4,6-trichlorobenzoyl chloride (TCBCl), also known as Yamaguchi reagent (Figure 1) is a reagent that is employed in an esterification process devised by Masaru Yamaguchi et al. in 1979 and is also effective in macrolactonization reactions. (https://doi.org/10.1246%2Fbcsj.52.1989).
Preparation
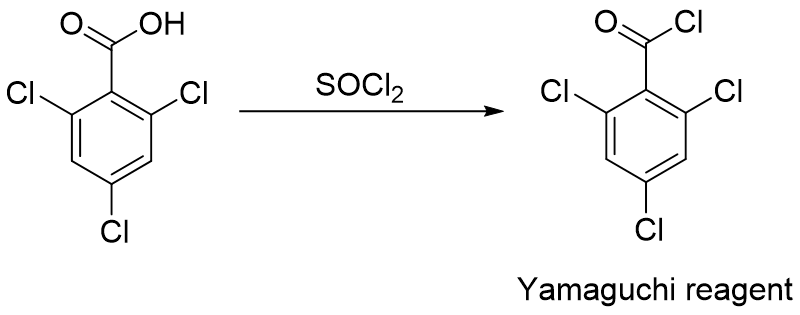
Yamaguchi reagent is commercially available, but it can also be made from slightly less expensive 2,4,6-trichlorobenzoic acid by utilizing chlorinating agents such as SOCl2 (Figure 2).
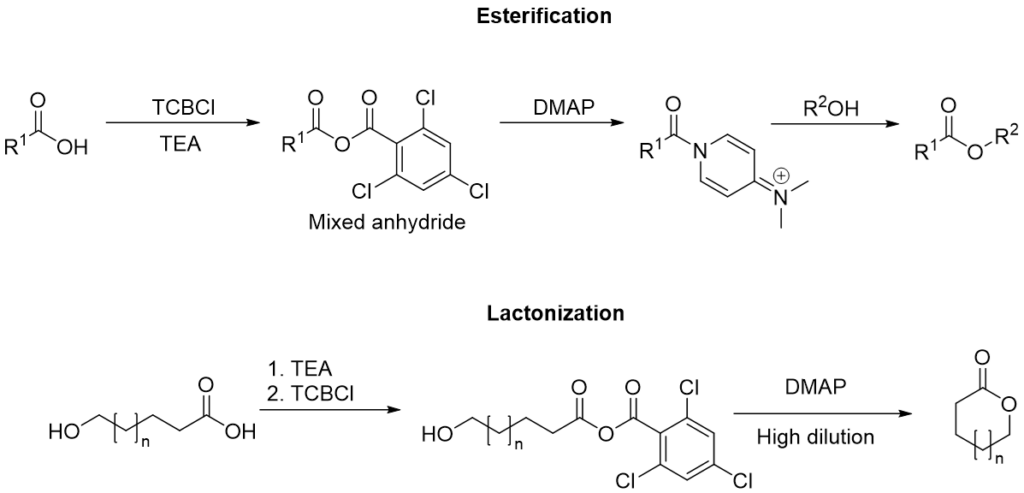
Yamaguchi esterification
In the original protocol for esterifications, the carboxylic acid is first treated with TCBCl and triethylamine (TEA), and after isolation of the mixed anhydride, the alcohol is added along with 4-dimethylaminopyridine (DMAP).
For lactonizations, the hydroxy acid is first deprotonated with TEA, and then treated with TCBCl, and after the removal of TEA.HCl from the mixture, the formed mixed anhydride is heated in toluene with DMAP under high dilution conditions to favour the intramolecular nucleophilic attack resulting in cyclization.
Application of Yamaguchi reagent in synthesis
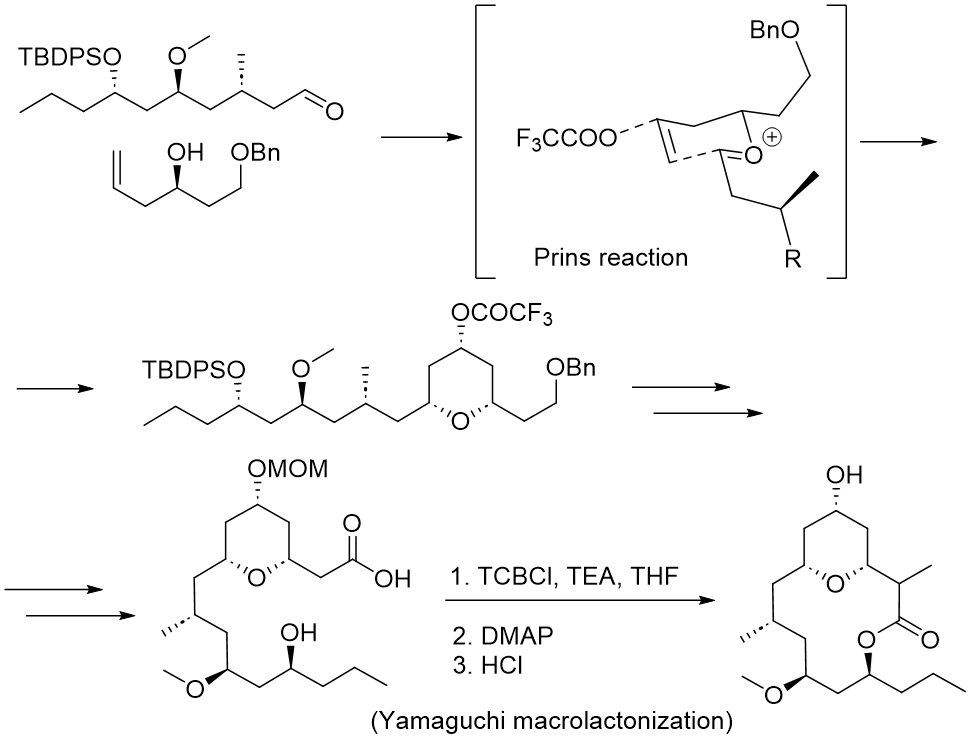
This approach was employed in a number of total syntheses of natural products containing a macrolactone motif and proven to be effective even for macrolactonizations.
Macrolides are one type of natural chemical. They are a family of natural compounds that comprise a macrolactone ring with deoxy sugar sidechains attached. Some macrolides have antibacterial activity, such as Erythromycin or Kitasamycin, whereas others have cytostatic activity, such as Neopeltolide (Figure 4).
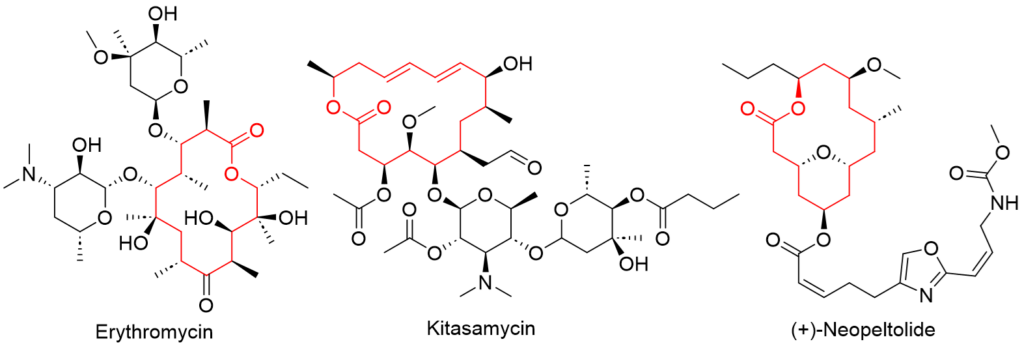
Finally, the Yamaguchi lactonization step from the 2008 total synthesis of Neopeltolide by Maier et al. (https://doi.org/10.1021/ol8001255) is illustrated as one of many applications of Yamaguchi esterification/lactonization in total syntheses.
If you are interested in organic reagents, you can find more articles here.
Santiago Lab is offering custom synthesis and contract research services in the fields of organic, bioorganic and medicinal chemistry.


